Sergio Castaneda Zegarra
In vivo mouse models to unravel the genetic interaction of immunodeficiencies
Sergio Castaneda Zegarra
Research Group: Oksenych Group
Department of Clinical and Molecular Medicine, Faculty of Medicine and Health Sciences

Our immune system is constantly fighting to protect us against pathogens and infections. For example, our adaptive immune system is constituted mainly by B and T lymphocytes. The development of these cells depends on a DNA repair pathway called Non-homologous DNA end joining (NHEJ). Consequently, when this mechanism fails, it might be reflected upon a reduced number of lymphocytes or even worse, a total absence of them, also called severe combined immunodeficiency (SCID). SCID patients are clinically prone to get infections, and one famous example that captured the world’s attention is the case of David Vetter. David was a child who was born in 1971 and died in 1984. He had SCID and lived all his life (12 years) in a plastic bubble free of pathogens.
During the last years, mutations in several NHEJ genes have been identified in humans. Most NHEJ patients possess SCID characteristics, including also neurological abnormalities. For instance, in 2006, Buck and colleagues reported five patients with mutations in XLF, which resulted in a new syndrome associated with immunodeficiency, growth retardation, and microcephaly. However, mutations in some other NHEJ genes have not yet been found in human immunodeficient patients up to the present. This is the case of the MRI (modulator of retroviral infection) gene, which has been recently studied during the last years by Valentyn Oksenych’s group in Norway and an American group.
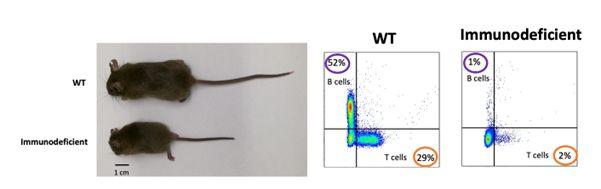
Similarly, SCID has been identified in NHEJ-deficient mice. Therefore, the use of transgenic mice has contributed to uncovering specific functions of DNA repair factors. The Oksenych’s group and their colleagues in the USA characterized two different MRI-deficient mice, concluding that MRI was largely dispensable for DNA repair and mouse development. However, our group (Oksenych’s group) has developed several new complex mouse models to study the immune system development when NHEJ is affected.
Our new models involve in vivo studies when one DNA repair factor is absent in combined deficiency with another NHEJ-related factor. For example, our team has shown that MRI is embryonically lethal when combined inactivation with two other NHEJ factors, rescuing one of them genetically and making possible its study. In contrast, another mouse model was viable and had almost normal B and T cell development when MRI was inactivated with another NHEJ factor. Our results have been successfully published in different scientific articles during the last years. My research project is a good example that in vivo mouse models contribute to scientific knowledge, generating new insights to understand what happens in humans under similar circumstances.
During my Ph.D. project at NTNU, I have developed several genetically modified mouse models that can be used to unravel secrets between the immune system and DNA repair. Eventually, and fundamentally, I expect that my research may contribute to early patient diagnosis in the future, with the main goal of improving their lives.
Main supervisor: Valentyn Oksenych
Project period: 2019-2022
