WP2
Norwegian Health Association Centre for Dementia Research
Work package 2: The initial entorhinal pathology and its underlying mechanisms in AD
About
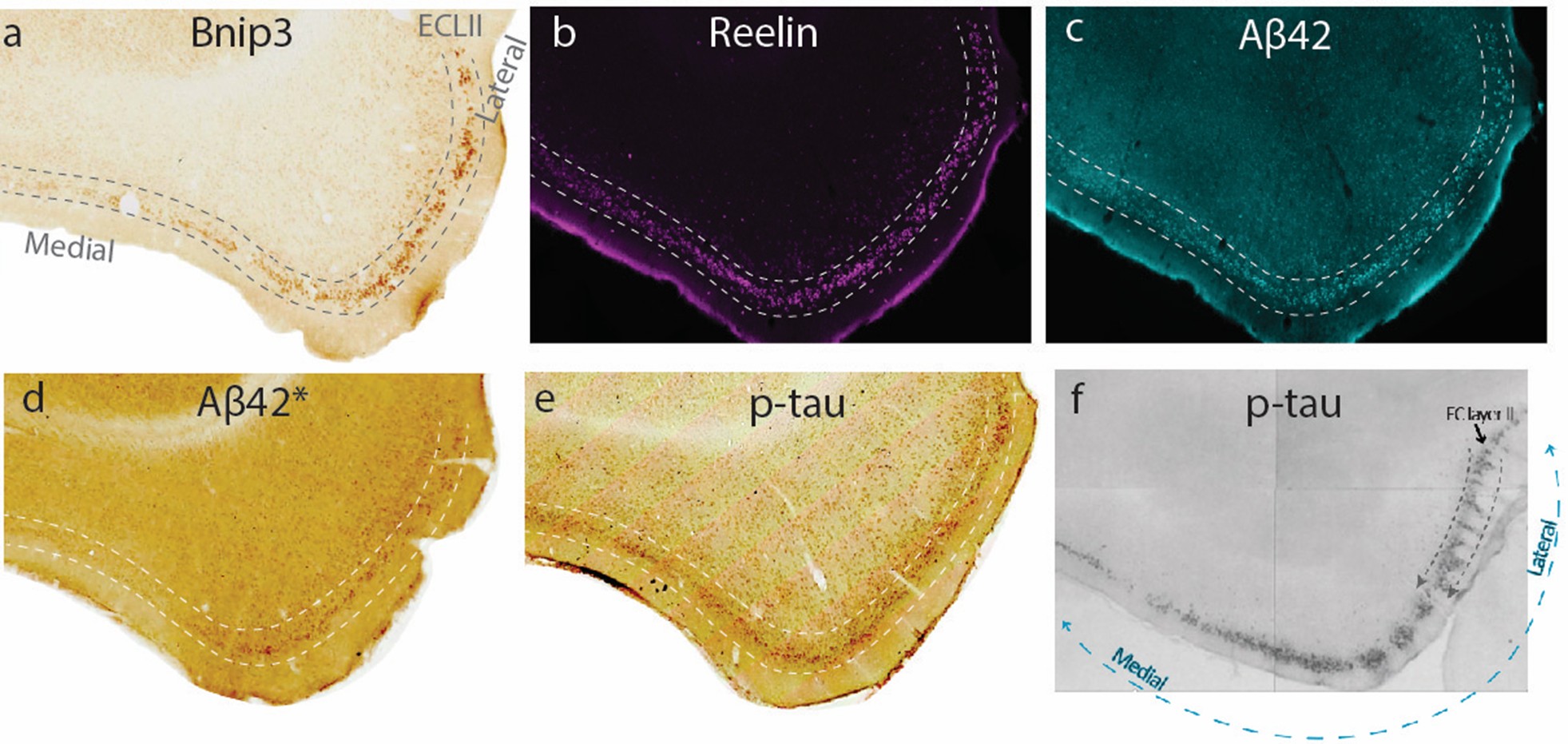
Since the brain contains certain populations of neurons that appear particularly vulnerable in the development of Alzheimer’s disease, we can probe these for clues that may help unlock fundamental mechanisms that trigger the disease. One such population consists of a group of big neurons located in a part of layer II of the lateral entorhinal cortex. These neurons develop pathology and start to die even before the appearance of clinical symptoms and may therefore constitute an origin point of the disease (Igarashi, 2023; Kobro-Flatmoen et al., 2021). An interesting feature of these lateral entorhinal layer II neurons is their unique combined expression of proteins, including two proteins called Bnip3 and Reelin (Omholt et al., 2024), that may help provide these neurons with the ability to quickly change their connectivity with other neurons. This feature could be related to the fact that these neurons are intimately involved in our memory capacities, including our ability to comprehend time. However, it may be that these very capabilities and the signaling pathways they require leave these neurons especially vulnerable to degenerative changes as we age.
This work package will therefore focus on whether age-related alterations in the neuronal environment give rise to pathological triggers that selectively impact the Bnip3- and Reelin expressing entorhinal layer II neurons due to their unique physiological signature.

WP researchers
-
Elly Isabella Hartong Engineer
elly.i.hartong@ntnu.no Kavli Institute for Systems Neuroscience -
Agalic Rodriguez Duboc Postdoctoral Fellow
agalic.rodriguez.duboc@ntnu.no Kavli Institute for Systems Neuroscience -
Sarah Judith Seidel PhD Candidate
sarah.j.seidel@ntnu.no Kavli Institute for Systems Neuroscience






