Ion mobility analysis
Ion mobility analysis
Ion mobility analysis
Ion-mobility spectrometry (IMS) is an analytical technique, mainly used in biomolecule analysis, with the basic aims of separating and identifying ionized molecules in the gas phase based on their electrical mobility in a carrier buffer gas. Electrical mobility is the ability of charged ions to move through a medium in response to an electric field that is applied on them. The separation of ions according to their mobility in a gas phase is based on differences of their shapes and charge states. A number of compounds (such as proteins) are rarely isolated in the solvent phase, but they are often stable in the gas phase, where ions from electrospray ionization can exist in varying charge states. Differences in the structures of molecules in the gas phase are often large enough to allow different shapes and charge states to be separated because of differences in their mobilities through a gas. Moreover, gentle collisional activation can be used to induce structural transformations of the target chemicals. These new structures often have different mobilities. Thus, there is the possibility of developing a multidimensional separation that takes advantage of structural differences of multiple stable states of a compound.
In the past ten years, combinations of IMS with MS and liquid chromatography (LC) have emerged as powerful, hyphenated platforms for examining complex molecular mixtures. Applications ranging from the high throughput detection of tens of thousands of peptide ions to the obtaining of structural insight about large protein complexes have stimulated significant interest by the scientific community. In the gas phase, molecules can display many nonnative chemical structures that are stable during the millisecond timescales of ion mobility experiments. Resolving different conformations is readily achieved and requires that some structures are not in equilibrium with one another. This behavior is different from the solvent-phase equilibrium of states that is responsible for the cooperative transitions normally observed in solvent. The ability to examine stable ion structures raises an opportunity to select specific ion shapes for activation and then examine the new structures that are formed in nondissociative collisions. The vacuum environment not only allows one to examine intramolecular interactions in the absence of solvent effects, it arguably may be the only place where it is possible to select many different types of precursors and intermediates in a way that allows the step-by-step delineation of transitions.
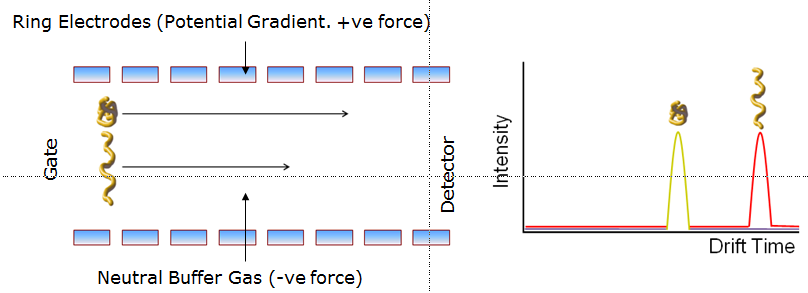
Reference: Brian C. Bohrer, Samuel I. Merenbloom, Stormy L. Koeniger,Amy E. Hilderbrand, David E. Clemmer, Biomolecule Analysis by Ion Mobility Spectrometry, Annu. Rev. Anal. Chem. 2008. 1:293–327.
