Polyelectrolyte complexes - Monte Carlo simulations of coarse-grained biophysical systems
MENU
Polyelectrolyte complexes
Polyelectrolyte complexes
– Monte Carlo simulations of coarse-grained biophysical systems
Polyelectrolyte complexes (PEC) are relevant in biological and technological processes. Chromatin, the DNA-histone protein complex that composes the nucleus of cells, is probably the most study biological "polyelectrolyte complex".
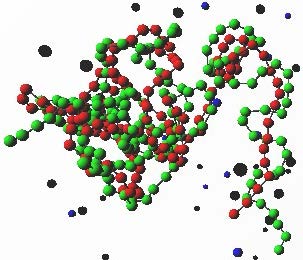 As an example of the relevance of PEC as technological applications we mention the large body of literature regarding DNA-oppositely charged polyelectrolyte complexes (polyplexes) for gene delivery applications. Here we use a simple bead-and-spring model to described the polyelectrolytes to study the impact of the properties of the polyelectrolytes, and co-solutes, on the topology and structure of the formed complexes.
As an example of the relevance of PEC as technological applications we mention the large body of literature regarding DNA-oppositely charged polyelectrolyte complexes (polyplexes) for gene delivery applications. Here we use a simple bead-and-spring model to described the polyelectrolytes to study the impact of the properties of the polyelectrolytes, and co-solutes, on the topology and structure of the formed complexes.
